This surge is driven by national immunization programs transitioning from cold-chain-dependent liquid vials and syringes to thermostable, self-administrable solid-dose transdermal platforms.
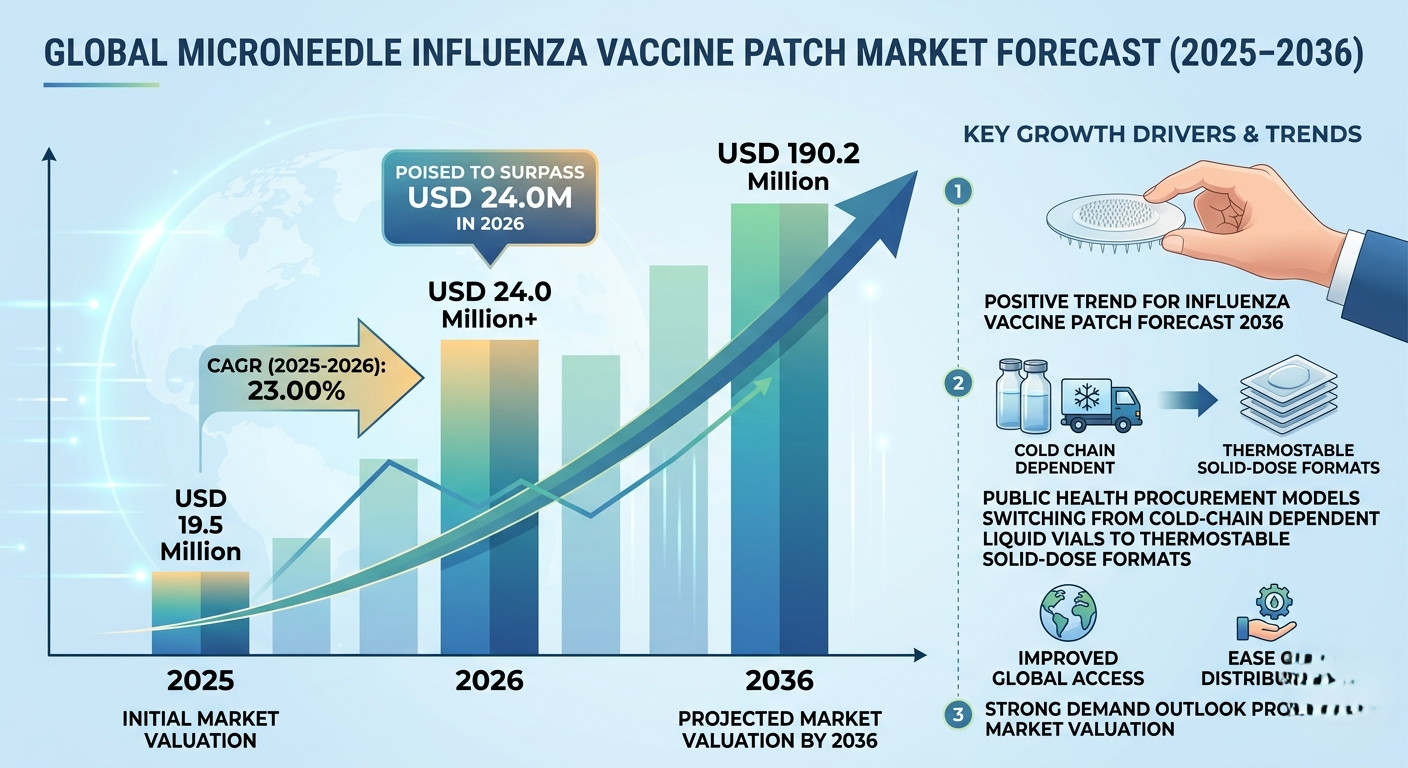
NEWARK, DE / ACCESS Newswire / March 25, 2026 / According to a new report by Future Market Insights (FMI) The global microneedle patch influenza vaccine delivery market is set for explosive growth, expanding from USD 24.0 million in 2026 to USD 190.2 million by 2036, registering a robust CAGR of 23.00% during the forecast period, according to the latest analysis by Future Market Insights (FMI).
This surge is driven by national immunization programs transitioning from cold-chain-dependent liquid vials and syringes to thermostable, self-administrable solid-dose transdermal platforms. The shift eliminates logistical friction around refrigerated transport, sharps disposal, and dependence on trained healthcare personnel-making rapid pandemic response and seasonal flu campaigns far more efficient.
Get Access of Report Sample: https://www.futuremarketinsights.com/reports/sample/rep-gb-32365
Microneedle Patch Influenza Vaccine Delivery Market Snapshot (2026-2036)
Market size in 2026: USD 24.0 million
Market size in 2036: USD 190.2 million
CAGR (2026-2036): 23.00%
Leading patch technology: High-density coated microarray patches (~34.8% share in 2026)
Leading vaccine format: Seasonal inactivated influenza vaccines (~46.2% share in 2026)
Leading administration model: Provider-administered clinic/pharmacy vaccination (~54.1% share in 2026)
Leading end user: Public health agencies and immunization programs (~40.7% share in 2026)
Dominant revenue stage: Clinical trial and pilot procurement (~61.5% share in 2026)
Fastest-growing countries: Australia (25.1%), United States (24.6%), United Kingdom (22.9%)
Market Momentum
The market, valued at USD 19.5 million in 2025, is poised to surpass USD 24.0 million in 2026 as early-stage clinical procurement, milestone payments, and pilot contracts dominate. By 2031, increasing public-sector preparedness procurement and initial commercial seasonal distributions will accelerate momentum. Between 2031 and 2036, the market is expected to scale dramatically as regulators authorize broader self-administration/mail-to-home models and national stockpiles fully adopt solid-state formats-reaching USD 190.2 million by 2036 at a sustained 23.00% CAGR.
Why the Market Is Growing
Public health agencies and biopharma developers are re-evaluating delivery infrastructure to address cold-chain vulnerabilities, healthcare workforce shortages, and the need for rapid pandemic deployment. Key drivers include:
Thermostability imperative: Solid-dose patches enable ambient storage and conventional mail distribution, decoupling vaccination rates from refrigerated logistics and trained personnel.
Operational efficiency: Elimination of sharps waste dramatically reduces backend logistics and biohazard costs.
Dose-sparing potential: High-density coated microarray patches target immune-rich epidermis (Langerhans cells), supporting profound antigen efficiency.
Pandemic preparedness: Formats compatible with inactivated, mRNA, or broadly protective antigens allow rapid strain updates and mail-to-home deployment.
Analyst OpinionSabyasachi Ghosh, Principal Analyst, Healthcare, at FMI, notes: "Public health agencies publicly celebrate microneedle patches for their pain-free patient appeal, but the actual procurement trigger is waste management. The elimination of millions of biohazardous sharps from the annual seasonal flu campaign changes the backend logistics cost so dramatically that governments are willing to subsidize the upfront device premium."
Segment Spotlight
Patch Technology: High-density coated microarray patches lead due to superior antigen preservation and dose-sparing without altering protein structure. Dissolving polymer and silk-based platforms offer waste elimination and extreme thermostability.
Vaccine Format: Seasonal inactivated vaccines dominate early volumes, providing scale for manufacturing lines; mRNA integration represents the next frontier for pandemic readiness.
Administration Model: Provider-administered settings serve as the critical bridge for real-world data before full self-administration authorization.
End User: Public health agencies provide the volume guarantees needed to de-risk continuous manufacturing scale-up.
Country Growth Outlook (CAGR 2026-2036)
Country |
CAGR |
|---|---|
Australia |
25.1% |
United States |
24.6% |
United Kingdom |
22.9% |
Germany |
22.4% |
Canada |
22.1% |
China |
21.8% |
India |
21.2% |
Australia leads thanks to its proactive national framework that rewards needle-free formats and leverages domestic microneedle IP. The United States follows closely, driven by massive seasonal demand and BARDA-backed rapid-response requirements.
Drivers, Opportunities, Trends, ChallengesDrivers:
Cold-chain elimination and sharps waste reduction
Healthcare workforce shortages pushing self-administration models
National security need for rapid pandemic countermeasures
Opportunities:
Direct-to-consumer pharmacy and mail-to-home distribution
Pediatric compliance and older-adult convenience differentiation
Premium integration with next-generation mRNA influenza vaccines
Trends:
Convergence of vaccine stabilization chemistry with micro-scale epidermal delivery
Shift from purchasing doses to acquiring complete delivery modalities
Licensing and exclusive device partnerships between biopharma and microneedle specialists
Challenges:
Absence of unified regulatory monographs for vaccine-device combination products
Bespoke validation pathways increasing time and cost for commercialization
Need to prove polymer clearance and long-term thermostability at scale
Competitive Landscape The market is intensely concentrated around proprietary microarray engineering patents and scalable manufacturing. Leading players are advancing clinical programs and securing partnerships for continuous production.
Key players include:
Vaxxas
Vaxess Technologies
Micron Biomedical
LTS Lohmann Therapie-Systeme AG
BioNet
Zipcode Bio
PopVax
Competition centers on demonstrated manufacturing scalability, IP strength, and compatibility with both seasonal inactivated and future mRNA antigens. Public buyers deliberately fund multiple architectures to avoid supply-chain lock-in.
Strategic Implications / Executive Takeaways
Biopharma developers should secure exclusive microneedle device partnerships to protect legacy antigens from commoditization.
Supply chain teams must pivot from cold storage expansion to high-throughput ambient networks.
Regulatory teams need to accelerate combination-product monographs to meet impending public tenders mandating solid-state capabilities.
For an in-depth analysis of evolving microneedle vaccine delivery trends and to access the complete strategic outlook for the Microneedle Patch Influenza Vaccine Delivery Market through 2036, visit the official report page at: https://www.futuremarketinsights.com/reports/microneedle-patch-influenza-vaccine-delivery-market
Explore More Research Reports by FMI:
Microneedle Drug Delivery Systems Market: https://www.futuremarketinsights.com/reports/microneedle-drug-delivery-systems-market
Seasonal Influenza Vaccines Therapeutics Market: https://www.futuremarketinsights.com/reports/seasonal-influenza-vaccines-therapeutics-market
COVID-19 Vaccine Packaging and Delivery Devices Market: https://www.futuremarketinsights.com/reports/covid-19-vaccine-packaging-and-delivery-devices-market
Vaccine Cryoprotectants Market: https://www.futuremarketinsights.com/reports/vaccine-cryoprotectants-market
Vaccine Residual Process Reagents Market: https://www.futuremarketinsights.com/reports/vaccine-residual-process-reagents-market
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
For Press & Corporate Inquiries
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - sales@futuremarketinsights.com
For Media - Rahul.singh@futuremarketinsights.com
For web - https://www.futuremarketinsights.com/
SOURCE: Future Market Insights, Inc.
View the original press release on ACCESS Newswire






